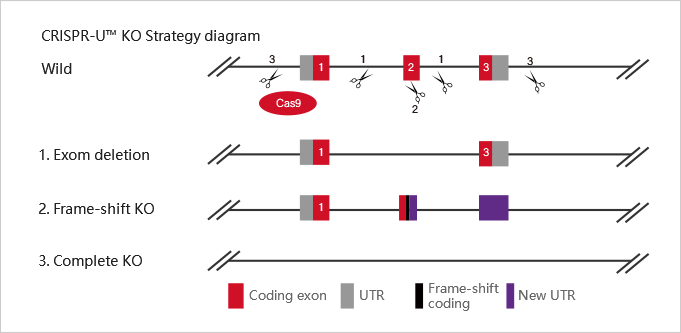
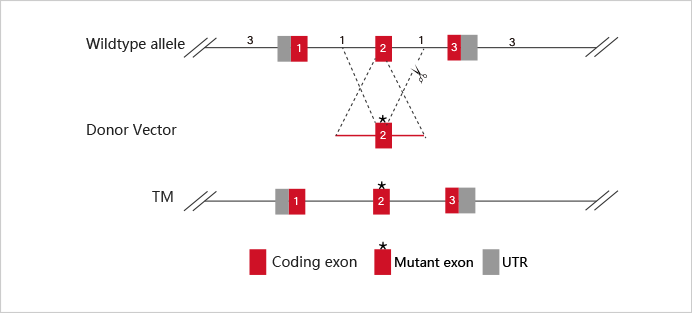
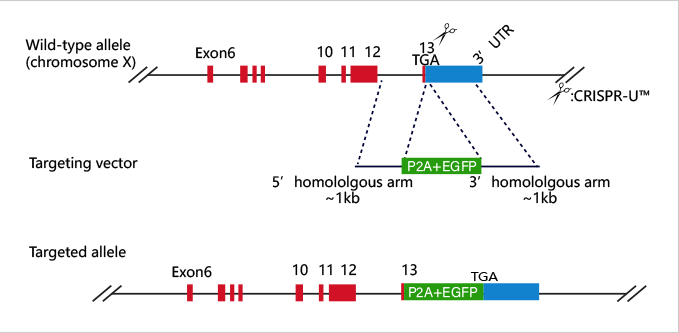
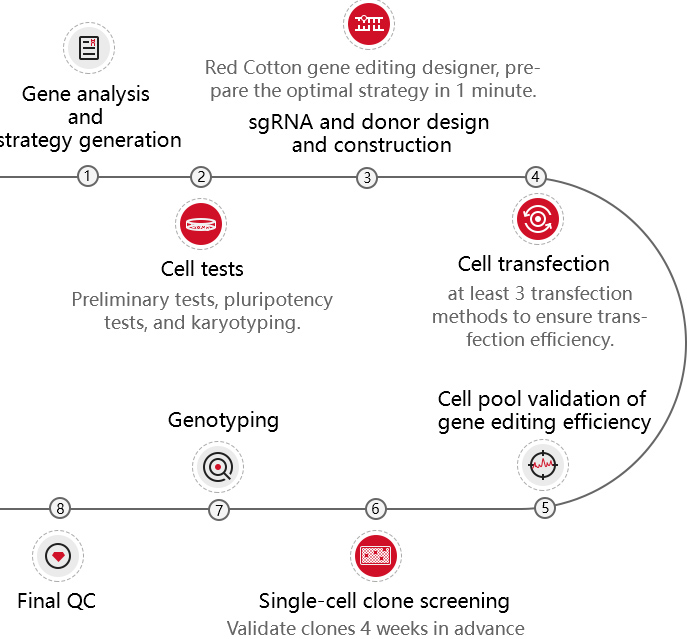
Embryonic stem cells (ESC) and induced pluripotent stem cells (iPSC) have pluripotency and can theoretically differentiate into all organs, tissues or cells. As early as the 1980s, mouse ESC has been applied to gene-recombination modification for gene function research. Specifically, it refers to transfecting the homologous targeting vector carrying modified gene into mouse ESCs, injecting the positive clones into blastocysts, and then transplanting them into surrogate mice to obtain chimeric mice. Human ESC is mainly applied in disease model construction. With the appearance of iPSC technology in 2006 and the development of more than ten years, iPSC has gradually replaced ESC in some researches.
At present, iPSC and ESC in the research of gene function, disease model, or drug screening usually needs to be combined with gene editing technology. CRISPR/Cas9, as the most mainstream gene editing technology in recent years, has the advantages of high efficiency, simple and low cost. It has been widely used in stem cells gene knockout, point mutation and knockin.
Ubigene's advantages
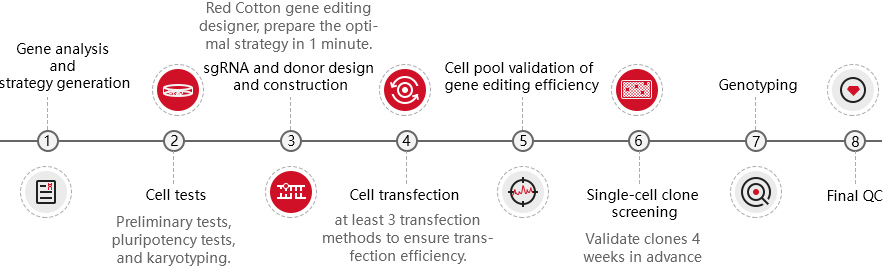
As a global company focusing on CRISPR gene editing technology in cell lines, Ubigene has comprehensively optimized the whole technical process of iPSC gene editing and successfully provided over a hundred gene-edited iPSC/ESC to more than 30 countries and regions in the world.
Patented CRIPSR-U™ technology
Highly efficient gene editing, positive rate elevated 10-20 times.
EZ-editor™ single-cell clone validation technology
High throughput positive clones screening, greatly shortening the project turnaround.
Stem cell culture system
Exclusive system to optimize cell condition and ensure cell pluripotency.
Rich experience
More than 200 cell lines, 5000 successful cases.
Case studies
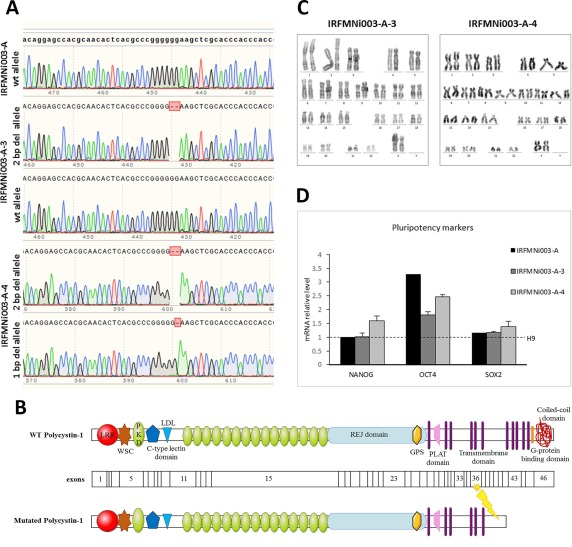
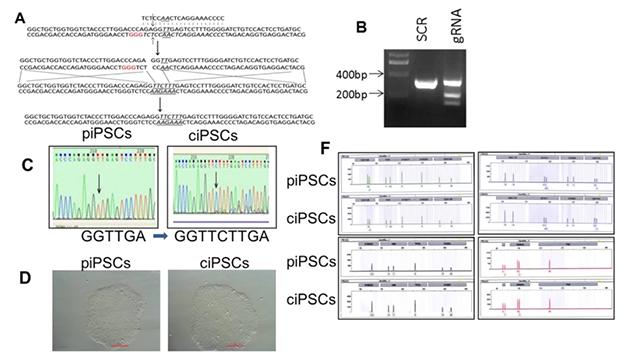
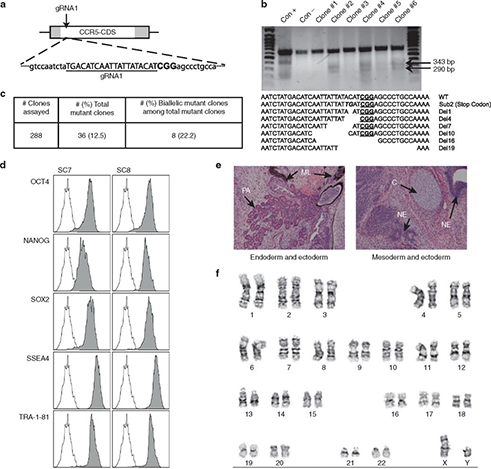
At present, there are many cases of CRISPR gene editing applied in iPSC for disease model construction, such as neurodegenerative diseases, metabolic diseases and cardiac genetic diseases. The following table summarizes some relevant studies in previous years.
Table 1: Cases of CRISPR gene editing applied in iPSC
| Gene editing type | Disease | Gene | Disease model | Cell therapy |
| CRISPR-KO disease modeling | Immunodeficiency, centromeric instability, and facial abnormalities (ICF) syndrome | DNMT3B | √ | |
| Dominant dystrophic epidermolysis bullosa(DDEB) | COL7A1 | √ | |
| Tangier disease (TD) | ABCA1 | √ | |
| Atrial fibrillation (AF) | KCNA5 | √ | |
| CRISPR-KO regain normal function | Fragile X syndrome (FXS) | FMR1 | √ | |
| Duchenne muscular dystrophy (DMD) | dystrophin | √ | √ |
| CRISPR-KI mutation repair | Myositis ossificans progressiva (MOP) | ALK-2 | √ | |
| Chronic granulomatous disease (CGD) | CYBB | √ | √ |
| Amyotrophic lateral sclerosis (ALS) | SOD1 and FUS | √ | |
| Frontotemporal dementia | CHMP2B | √ | |
| Abetalipoproteinemia | MTTP | √ | |
| Hypertrophic cardiomyopathy (HCM) | PRKAG2 | √ | |
| CRISPR combined with piggyBAC | Beta-thalassemia | HBB | √ | √ |
| Brugada syndrome | SCN5A | √ | |
| Huntington's disease(HD) | HTT | √ | |
| Hereditary motor sensory neuropathy (HMSN) | TFG | √ | |
| Tetrahydrobiopterin (BH4) deficiency | PTPS and DHPR | √ | |
| HIV | CCR5 | √ | √ |
| Polycythemia vera (PV), α 1-antitrypsin (AA T) deficiency | JAK2-V617F and SERPINA1 | √ | |
| CRISPRi gene knockdown | Long QT syndrome(LQTS) | CALM2 | √ | |