Location:Home > Application > Kir2.1 overexpression helps to study new therapeutic target for vascular remodelling diseases
[Research highlight] Kir2.1 channel could be a new target for vascular remodeling diseases treatment
Background
Bone marrow derived endothelial progenitor cells (EPCs) can be mobilized, homed to the site of local vascular injury and differentiated into vascular endothelial cells in vascular repair. Therefore, EPCs are the critical cells of postnatal neovascularization and angiogenesis. Accumulating evidence and previous reports have shown the potential of treatment with EPC transplantation in animal models of carotid artery injury and hind limb and myocardial ischaemia. The recent literature has shown that under certain physiological or pathophysiological conditions, EPCs can undergo an endothelial-mesenchymal transition (EndoMT); Thus, their function will presumably be altered. However, the underlying mechanism of this transformation process has not yet been clarified. Recent research also showed that the Kir2.1 channel is closely related to the differentiation of EPCs. However, whether alterations in Kir2.1 channel expression affect the differentiation (transdifferentiation) or subsequent cytological functions of EPCs has not been discussed.
In response to this question, the researchers from Institute of Stem Cell and Regenerative Medicine in Medical College of Qingdao University published an article named “Slight up-regulation of Kir2.1 channel promotes endothelial progenitor cells to transdifferentiate into a pericyte phenotype by Akt/mTOR/Snail pathway” on JOURNAL OF CELLULAR AND MOLECULAR MEDICINE. They reported that the slight up-regulation of Kir2.1 promoted channel hyperpolarization, contributing to EPC mesenchymal transition, and that this process has an essential function in angiogenesis and vascular stability.

In this study, the researchers found that the slight up-regulation of Kir2.1 expression promoted the expression of the stem cell stemness factors ZFX and NS and inhibited the expression of senescence-associated β-galactosidase. Further studies showed the slightly increased expression of Kir2.1 could also improve the expression of pericyte molecular markers NG2, PDGFRβ and Desmin. Moreover, adenovirus-mediated Kir2.1 overexpression had an enhanced contractile response to norepinephrine of EPCs. These results suggest that the up-regulated expression of the Kir2.1 channel promotes EPCs transdifferentiation into a pericyte phenotype. Furthermore, the mechanism of EPC transdifferentiation to mesenchymal cells (pericytes) was found to be closely related to the channel functional activity of Kir2.1 and revealed that this channel could promote EPC EndoMT by activating the Akt/mTOR/Snail signalling pathway.
EPC-derived pericytes express the pericyte marker protein Desmin and promote the maturation and stabilization of neovascularization
Desmin is a structural and functional protein of pericytes that is important for the maintenance of vascular stability and pericyte contraction. The results of immunofluorescence assays showed that the protein expression of Desmin was significantly increased when EPCs were transfected with pAV-Kir2.1 (Figure 1A). To observe the effect of transdifferentiated EPCs on the stability of vascular endothelial cells, the transformed EPCs were cocultured with RAECs (provided by Ubigene) in Matrigel matrix. Transdifferentiated EPCs exhibited longer vascular tubes in vitro (Figure 1B & C) and a higher capillary-like structure maintenance rate (Figure 2) than the control group, which indicates that Kir2.1 overexpression in EPCs can promote angiogenesis and stabilize neovascularization. In this study, Ubigene provided CM-Dil-labelled RAECs to observe and analyze tube formation under the fluorescence microscope.
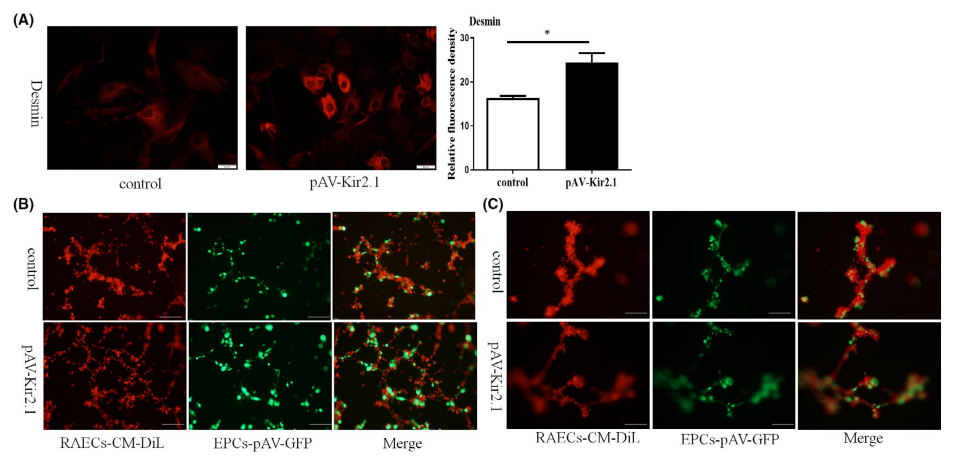
Figure 1
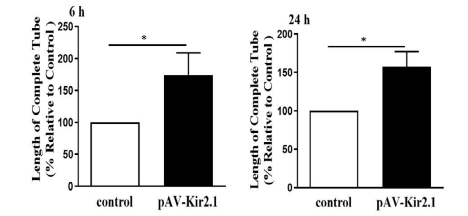
Figure 2
The slightly up-regulated expression of Kir2.1 promotes EPCs transdifferentiation into a pericyte phenotype by the Akt/mTOR/Snail signalling pathway
They further investigated the role of potential molecular signalling pathways in Kir2.1-mediated EPCs transdifferentiation. It was found that increased expression of Kir2.1 enhanced the level of intracellular Akt and mTOR phosphorylation and promoted expression of the Snail protein. Treatment with the Kir2.1 channel blocker ML133 abolished these effects (Figure 3A). Next, the Akt phosphorylation blocker MK2206, the p-mTOR inhibitor RAD001 and Snail siRNA were applied to inhibit or interfere with the expression of the corresponding signal molecules. Then, the effects of these changes on expression of the marker molecules NG2 and PDGFRβ and the stem stemness factors ZFX and NS were detected. The results showed that MK2206, RAD001 and Snail siRNA could attenuate the up-regulated expression of NG2, PDGFRβ, NS and ZFX promoted by Kir2.1 (Figure 3B), suggesting that Akt/mTOR/Snail plays a central role in the EndoMT of Kir2.1-induced EPCs. In summary, these results suggest that slight Kir2.1 overexpression could promote EPCs transdifferentiation through the Akt/mTOR/Snail pathway.
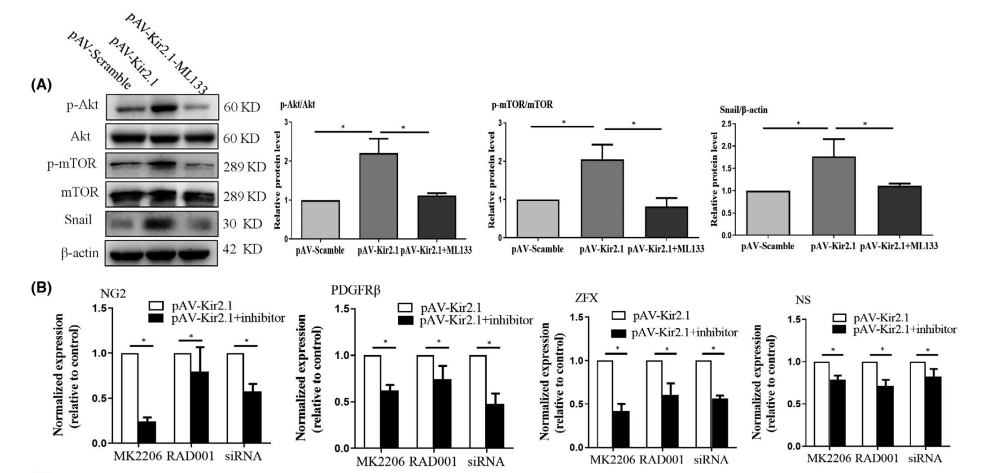
Figure 3
Overall, this study suggested that in the early stage of inflammatory response, regulating the Kir2.1 channel expression affects the biological function of EPCs, thereby determining the maturation and stability of neovascularization. And the researchers speculated that the Kir2.1 channel is likely a switch molecule by which EPCs perceive the stem niche and thus determine the direction of EPCs differentiation or transdifferentiation, and Kir2.1 may be a new therapeutic target for vascular remodelling diseases.
Ubigene is an enterprise focusing on gene editing cell line. We provide high-quality gene-editing cell lines, stable cell lines, virus packaging and other related services, as well as nearly 2000 KO cell lines. Please contact us for more details!
[Research highlight] Kir2.1 channel could be a new target for vascular remodeling diseases treatment

Background
Bone marrow derived endothelial progenitor cells (EPCs) can be mobilized, homed to the site of local vascular injury and differentiated into vascular endothelial cells in vascular repair. Therefore, EPCs are the critical cells of postnatal neovascularization and angiogenesis. Accumulating evidence and previous reports have shown the potential of treatment with EPC transplantation in animal models of carotid artery injury and hind limb and myocardial ischaemia. The recent literature has shown that under certain physiological or pathophysiological conditions, EPCs can undergo an endothelial-mesenchymal transition (EndoMT); Thus, their function will presumably be altered. However, the underlying mechanism of this transformation process has not yet been clarified. Recent research also showed that the Kir2.1 channel is closely related to the differentiation of EPCs. However, whether alterations in Kir2.1 channel expression affect the differentiation (transdifferentiation) or subsequent cytological functions of EPCs has not been discussed.
In response to this question, the researchers from Institute of Stem Cell and Regenerative Medicine in Medical College of Qingdao University published an article named “Slight up-regulation of Kir2.1 channel promotes endothelial progenitor cells to transdifferentiate into a pericyte phenotype by Akt/mTOR/Snail pathway” on JOURNAL OF CELLULAR AND MOLECULAR MEDICINE. They reported that the slight up-regulation of Kir2.1 promoted channel hyperpolarization, contributing to EPC mesenchymal transition, and that this process has an essential function in angiogenesis and vascular stability.

In this study, the researchers found that the slight up-regulation of Kir2.1 expression promoted the expression of the stem cell stemness factors ZFX and NS and inhibited the expression of senescence-associated β-galactosidase. Further studies showed the slightly increased expression of Kir2.1 could also improve the expression of pericyte molecular markers NG2, PDGFRβ and Desmin. Moreover, adenovirus-mediated Kir2.1 overexpression had an enhanced contractile response to norepinephrine of EPCs. These results suggest that the up-regulated expression of the Kir2.1 channel promotes EPCs transdifferentiation into a pericyte phenotype. Furthermore, the mechanism of EPC transdifferentiation to mesenchymal cells (pericytes) was found to be closely related to the channel functional activity of Kir2.1 and revealed that this channel could promote EPC EndoMT by activating the Akt/mTOR/Snail signalling pathway.
EPC-derived pericytes express the pericyte marker protein Desmin and promote the maturation and stabilization of neovascularization
Desmin is a structural and functional protein of pericytes that is important for the maintenance of vascular stability and pericyte contraction. The results of immunofluorescence assays showed that the protein expression of Desmin was significantly increased when EPCs were transfected with pAV-Kir2.1 (Figure 1A). To observe the effect of transdifferentiated EPCs on the stability of vascular endothelial cells, the transformed EPCs were cocultured with RAECs (provided by Ubigene) in Matrigel matrix. Transdifferentiated EPCs exhibited longer vascular tubes in vitro (Figure 1B & C) and a higher capillary-like structure maintenance rate (Figure 2) than the control group, which indicates that Kir2.1 overexpression in EPCs can promote angiogenesis and stabilize neovascularization. In this study, Ubigene provided CM-Dil-labelled RAECs to observe and analyze tube formation under the fluorescence microscope.

Figure 1

Figure 2
The slightly up-regulated expression of Kir2.1 promotes EPCs transdifferentiation into a pericyte phenotype by the Akt/mTOR/Snail signalling pathway
They further investigated the role of potential molecular signalling pathways in Kir2.1-mediated EPCs transdifferentiation. It was found that increased expression of Kir2.1 enhanced the level of intracellular Akt and mTOR phosphorylation and promoted expression of the Snail protein. Treatment with the Kir2.1 channel blocker ML133 abolished these effects (Figure 3A). Next, the Akt phosphorylation blocker MK2206, the p-mTOR inhibitor RAD001 and Snail siRNA were applied to inhibit or interfere with the expression of the corresponding signal molecules. Then, the effects of these changes on expression of the marker molecules NG2 and PDGFRβ and the stem stemness factors ZFX and NS were detected. The results showed that MK2206, RAD001 and Snail siRNA could attenuate the up-regulated expression of NG2, PDGFRβ, NS and ZFX promoted by Kir2.1 (Figure 3B), suggesting that Akt/mTOR/Snail plays a central role in the EndoMT of Kir2.1-induced EPCs. In summary, these results suggest that slight Kir2.1 overexpression could promote EPCs transdifferentiation through the Akt/mTOR/Snail pathway.

Figure 3
Overall, this study suggested that in the early stage of inflammatory response, regulating the Kir2.1 channel expression affects the biological function of EPCs, thereby determining the maturation and stability of neovascularization. And the researchers speculated that the Kir2.1 channel is likely a switch molecule by which EPCs perceive the stem niche and thus determine the direction of EPCs differentiation or transdifferentiation, and Kir2.1 may be a new therapeutic target for vascular remodelling diseases.
Ubigene is an enterprise focusing on gene editing cell line. We provide high-quality gene-editing cell lines, stable cell lines, virus packaging and other related services, as well as nearly 2000 KO cell lines. Please contact us for more details!
