Expert Insights | Stable Cell Construction: Lentivirus VS Transposon
In life sciences research, the construction of stable cell lines (stable expression) is a very
important technical method. It provides a solid foundation for in-depth exploration of gene function, disease
mechanisms, and drug development. Currently, common methods for constructing stable cell lines include lentivirus,
transposons, CRISPR-Cas9 gene knock-in, and plasmid transfection. These methods each have their own strengths,
providing researchers with diverse choices. Today, follow Ubigene to explore the application scenarios and
characteristics of lentivirus and transposon methods.
Construction of
Lentivirus-Mediated Stable Cell Lines
Lentivirus, as a powerful gene vector, relies on its strong infectivity and efficient gene
integration characteristics, making it the mainstream method for constructing stable cell lines.
1.1Principle
Lentivirus belongs to the retrovirus family, capable of integrating
the exogenous gene it carries into the host cell genome, thereby achieving long-term stable gene expression.
Lentiviral vectors typically consist of packaging plasmids, envelope plasmids, and transfer plasmids. By
co-transfecting these three plasmids into packaging cells, infectious lentiviral particles can be produced.
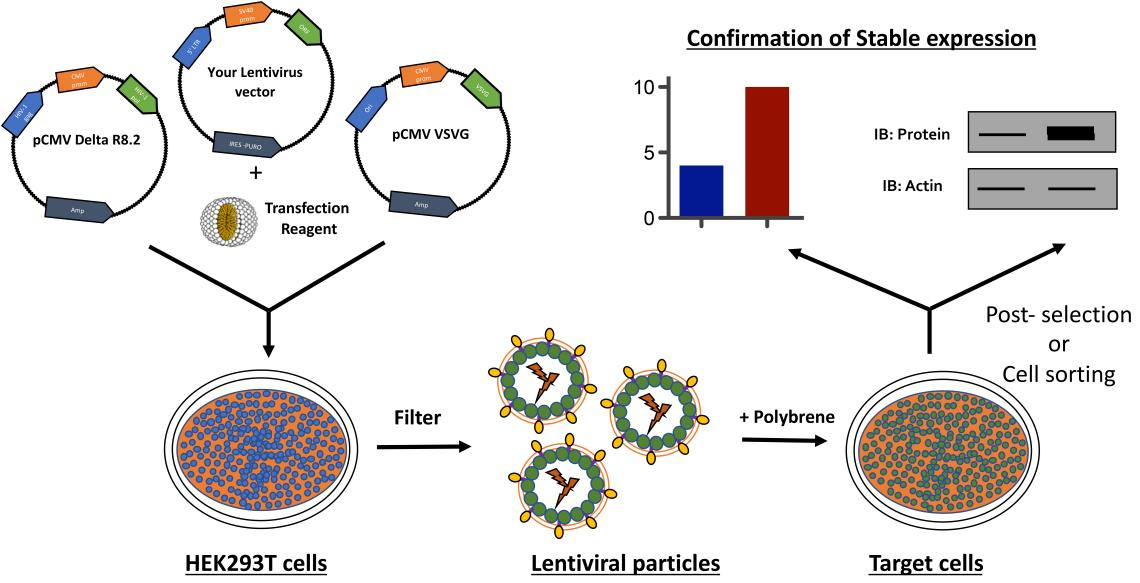
Figure 1 Schematic diagram of constructing stable cell lines using
lentivirus
1.2Construction Process
Vector Construction: Clone or synthesize the target gene and clone it into an appropriate
lentiviral vector.
Viral Packaging: Co-transfect the constructed lentiviral vector containing the target gene with
packaging plasmids into packaging cells such as 293T. Culture the cells and collect the supernatant containing the
lentiviral particles.
Viral Titer Determination: Determine the titer of lentivirus using appropriate methods (such as
qPCR or fluorescence assays).
Transduction of Target Cells: Seed the cells to be infected into culture plates and culture
them to an appropriate confluency or density. Based on
pre-experimental determination of the multiplicity of infection (MOI), add an appropriate amount of lentiviral
particles for infection. During infection, polybrene can be added depending on the cell type and virus
characteristics to enhance infection efficiency.
Selection of Stable Cells: 48-72 hours post-infection, add the appropriate concentration of
selection drugs (such as puromycin, zeocin, etc.) based on the selection marker used. Continue culturing and replace
the medium containing the selection drug until all uninfected cells die, leaving surviving cells as preliminarily
selected stable cell lines.
Cloning (Optional): Perform further single-cell cloning using the
selected cells by limited dilution or flow
cytometry to obtain genetically homogeneous stable cell lines.
Validation: Validate the integration and expression of the exogenous gene using methods such as
PCR, Western blot, etc.
Storage and Expansion: Freeze and store
the selected stable cell lines, and expand them as needed.
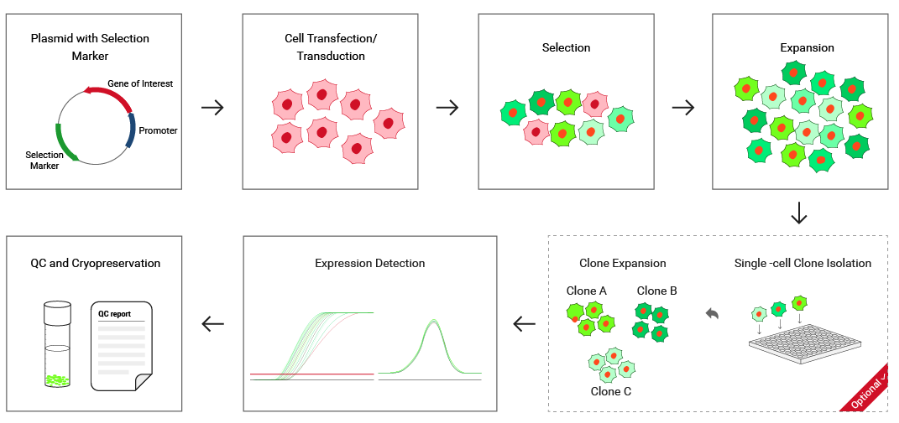
Figure 2 Ubigene's Stable Cell
Construction Service Workflow
1.3Application Scenarios
Cancer Research: In cancer research, lentivirus is commonly used to construct cell lines stably
expressing tumor suppressor genes or oncogenes, studying their effects on biological behaviors such as
proliferation, apoptosis, and migration of tumor cells.
Neuroscience: Due to the fact that neurons and other nerve cells are terminally differentiated
cells, conventional transfection methods are ineffective, while lentivirus can effectively infect these cells,
providing powerful tools for neuroscience research.
Immune Cell Research: For immune cells such as T cells, macrophages, etc., lentivirus can also
achieve efficient gene transduction, aiding research on the activation, differentiation, and immune response
processes of immune cells.
Construction of
Transposon-Mediated Stable Cell Lines
Transposons, as DNA sequences capable of jumping within the genome,
provide another unique strategy
for constructing stable cell lines.
1.1Working Mechanism
Transposons are a class of DNA fragments that can move within the genome, catalyzed by their encoded
transposase, from one location to another. When constructing stable cell lines, the exogenous gene is typically
inserted into a transposon vector, and then integrated into the host cell genome through the action of the
transposase.
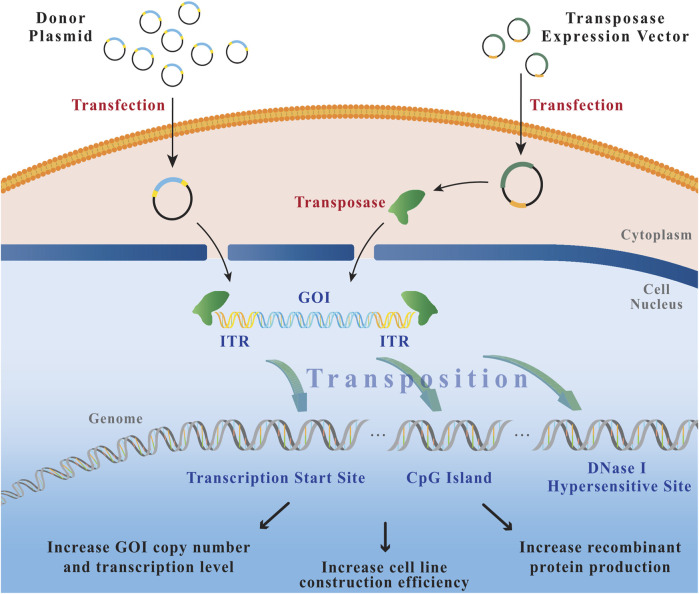
Figure 3
Transposition process
1.2Construction Process
Transposon Plasmid Construction: Insert the target gene into the transposon plasmid, usually
adding inverted terminal repeats (ITRs) of the transposon on either side of the target gene.
Co-transfection: Co-transfect the transposon plasmid with a helper plasmid
encoding the transposase into the target cells. Transfection
methods such as lipofection, electroporation, etc., can be used.
Selection of Stable Cell Lines: After transfection, transposition events occur in the cells,
integrating the target gene into the genome. Through appropriate selection methods (such as antibiotic selection),
select cells with stable integration of the target gene.
Cloning (Optional): Perform further single-cell cloning using the
selected cells by limited dilution or flow
cytometry to obtain genetically homogeneous stable cell lines.
Validation: Identify the selected stable cell lines, such as by verifying the integration of
the target gene through PCR, Southern blot, and evaluating the expression level of the target gene through Western
blot, fluorescence microscopy, etc.
Storage and Expansion: Freeze and store
the selected stable cell lines, and expand them as needed.
1.3Application Scenarios
Large Gene Sequence: Due to its
advantage of carrying larger payloads, transposons can accommodate larger genes without the need for cDNA
truncation. While lentiviral vector has limitations on the size of the
inserted gene.
For cells sensitive to lentivirus: If cells are sensitive to viral infection, lentiviral
methods may have a negative impact on cell survival, function, and overall condition. In such cases, consider using
transposons.
Below is a table summarized by Ubigene for reference:
|
Lentivirus
|
Transposon (e.g., PiggyBac)
|
Gene Capacity
| 6-6.5kb | 10-30kb |
Integration Site
|
Random integration into host cell genome
|
Specific integration into TTAA sites
|
Applicable Cell Types
|
Broad, including dividing and non-dividing cells
|
Relatively broad, but may require optimization of transfection methods for different cell types
|
Features
|
Requires virus packaging and purification, complex process
|
Relatively simple, achieved through plasmid co-transfection
|
Summary
Lentivirus and transposons each have their advantages and
applications in stable cell line construction, but both suffer from random integration and uncertain copy numbers.
CRISPR-Cas9 gene knock-in can serve as an important alternative, but it is costly and time-consuming to produce.
Ubigene's EZ-OE™ technology offers a new, more stable, and reliable solution for stable cell line construction. This
technology differs from traditional transposon methods by enabling precise genome integration and single-copy
insertion, with low cost and a quick turnaround as fast as 5 weeks!
Tandon, N., Thakkar, K. N., LaGory, E. L., Liu, Y. and Giaccia, A. J. (2018). Generation of Stable
Expression Mammalian Cell Lines Using Lentivirus. Bio-protocol 8(21): e3073.
Sato M, Inada E, Saitoh I, Watanabe S, Nakamura S. piggyBac-Based Non-Viral In Vivo Gene Delivery
Useful for Production of Genetically Modified Animals and Organs. Pharmaceutics. 2020 Mar 19;12(3):277.
Wei, Mian et al. “Progress of Transposon Vector System for Production of Recombinant Therapeutic
Proteins in Mammalian Cells.” Frontiers in bioengineering and biotechnology vol. 10 879222. 4 May. 2022,
doi:10.3389/fbioe.2022.879222











